This article is a popular-science adaptation based on the scientific review “Cajal’s organization of neuronal nucleus revisited” (2025). We wish to express our sincerest gratitude to the authors: Miguel Lafarga, María T. Berciano, J. Oriol Narcis, Fernando C. Baltanás, and Olga Tapia and to the editor Fernando de Castro, for their extraordinary research work that keeps Cajal’s scientific legacy alive. We also extend special thanks to Estanislao Nistal for his valuable critical review of this text.
1. Historical Preamble and Scientific Contextualisation: The “Third Element” of the Neuron
The history of neuroscience rests upon pillars that are frequently perceived as immovable and completely defined. Among them, the figure of Santiago Ramón y Cajal stands with indisputable monumentality, primarily associated with the formulation of the Neuron Doctrine and the exhaustive analysis of synaptic connectivity and tissue architecture of the nervous system. However, there is a dimension of Cajal’s work, often eclipsed by his descriptions of axons and dendrites, that constitutes an intellectual feat of equal magnitude: his pioneering exploration of the internal architecture of the cell nucleus.
In the context of the celebrations of the recently concluded “Santiago Ramón y Cajal Research Year”, extended to 2025 by the Government of Spain as an Event of Exceptional Public Interest, it is imperative to re-examine this facet of the Nobel laureate. The recent review published by the group of Miguel Lafarga, María T. Berciano, and collaborators, titled “Cajal’s organization of neuronal nucleus revisited” (2025), provides the perfect platform for this analysis.
At the beginning of the twentieth century, cell biology was in an embryonic stage, constrained by the resolution of optical microscopes and the availability of rudimentary staining techniques. While the prevailing dogma, influenced by Golgi’s reticular theory, visualised the nervous system as a continuous network, Cajal demonstrated the individuality of nerve cells. But Cajal did not stop at the plasma membrane. His curiosity led him to probe the interior of the soma, confronting the terra incognita of the neuronal nucleus. In 1910, Cajal published a seminal work: “The Nucleus of the Pyramidal Cells of the Human Brain and of Certain Mammals” (“El núcleo de las células piramidales del cerebro humano y de algunos mamíferos”). In this text, Cajal applied his observational genius not to the external ramifications but to the “nuclear geography”, describing with an almost prophetic precision a series of substructures that today, more than a century later, are recognised as fundamental for gene expression and cellular homeostasis.
The present article aims to break down, expand, and critically analyse the findings presented in the recent scientific literature, integrating them with a vast supporting base spanning from protein biochemistry to the molecular pathology of neurodegenerative diseases. It will be demonstrated how Cajal’s morphological descriptions — based on silver affinity — anticipated the existence of membraneless organelles formed by liquid-liquid phase separation (LLPS), and how the dysfunction of these structures, which Cajal drew with India ink and infinite patience, underlies devastating pathologies such as Amyotrophic Lateral Sclerosis (ALS) and Spinal Muscular Atrophy (SMA).
This article is not merely historiographic; it is a vindication of morphology as a tool for functional discovery. By revisiting the “argyrophilic spherules”, the “hyaline clumps”, and the “accessory body”, we not only validate Cajal’s eye but find keys to understanding the most complex molecular mechanisms of the eukaryotic cell.
2. Methodology and Physico-Chemical Foundations: The Alchemy of Silver and Nuclear Matter
To understand the depth of Cajal’s findings, it is essential to dissect the tool that made them possible: the reduced silver nitrate method. Unlike the conventional basophilic stains of the era (such as Nissl’s methylene blue), which revealed nucleic acids in a general manner, Cajal’s silver technique offered a unique chemical specificity that, paradoxically, depended on physical properties of proteins that we have only begun to understand in the last decade.
2.1 The Reduced Silver Nitrate Procedure: Beyond Staining
Developed and perfected by Cajal in 1903, this method represented a quantum leap in histology. While Golgi’s method (“Reazione Nera”) produced a stochastic and complete impregnation of the neuron, useful for external morphology, Cajal’s reduced silver method allowed the visualisation of the cytoskeleton (neurofibrils) and, crucially, specific compartments within the nucleoplasm.
The protocol involved tissue fixation (usually with ammoniacal alcohol or formalin), followed by impregnation in a silver nitrate solution (AgNO3) and a subsequent chemical reduction using agents such as hydroquinone or pyrogallol. The key to Cajal’s success lay in his ability to manipulate fixation and reduction conditions, achieving the highlighting of specific nuclear structures based on their differential argyrophilia.
Analysis of historical documents and technical literature reveals that silver affinity is not uniform. Cajal categorised nuclear structures by the intensity and tone of the argentic precipitation:
-
Intense Argyrophilia (Black/Sepia Colour): Indicative of a high density of silver nucleation sites. This category includes the nucleolus (specifically its fibrillar components) and the accessory body (Cajal Body), which appeared as black, sharp, and dense structures against a lighter background.
-
Moderate Argyrophilia (Yellow/Red/Brown Colour): Observed in the hyaline clumps (nuclear speckles). This paler staining suggests a different chemical composition or lower molecular compaction density.
-
Punctate Argyrophilia: Characteristic of the neutrophil granules (transcription factories), which appeared as tiny dispersed black dots.
2.2 Biochemical Bases of Argyrophilia: The Connection with Acidic Residues and Phosphorylation
For decades, the exact mechanism of Cajal’s silver stain was a “black art”, empirical and poorly understood. However, modern biochemistry has revealed that nuclear argyrophilia resides in the presence of specific proteins with very defined molecular characteristics. Silver ions (Ag+) interact preferentially with negatively charged chemical groups, which act as “seeds” for the reduction to metallic silver (Ag0).
Current research identifies two main molecular determinants for this reaction in the nucleus:
-
Acidic Amino Acid Residues: Proteins rich in aspartic acid (Asp) and glutamic acid (Glu). The side-chain carboxyl groups (COO-) of these amino acids coordinate silver ions.
-
Phosphorylation: The nucleus is a centre of intense kinase activity. Nuclear proteins, especially those involved in transcriptional regulation and RNA processing, are frequently hyperphosphorylated. Phosphate groups (PO43-) offer high-affinity sites for silver.
The proteins that Cajal was visualising unknowingly include classical nuclear markers now well characterised:
-
Nucleolin (C23): A multifunctional nucleolar phosphoprotein, extremely rich in acidic stretches.
-
Nucleophosmin (B23): A molecular chaperone involved in ribosomal assembly.
-
UBF (Upstream Binding Factor): An rDNA transcription factor, crucial in the nucleolar fibrillar centres.
-
Coilin: The scaffolding protein of the Cajal Body.
It is remarkable that Cajal’s technique was able to discriminate these proteins in a complex cellular environment, acting as a “primitive immunocytochemistry” based on physicochemical properties.
2.3 From Argyrophilia to Fluid Physics: Intrinsically Disordered Regions (IDRs) and Phase Separation (LLPS)
One of the most profound revelations emerging from the comparison between Cajal’s drawings and twenty-first-century molecular biology is the relationship between argyrophilia and Intrinsically Disordered Regions (IDRs). The current literature explicitly highlights that most of the nuclear components described by Cajal are now recognised as “nuclear condensates” assembled through mechanisms of liquid-liquid phase separation (LLPS).
The proteins driving LLPS (such as Fibrillarin, Nucleolin, SRRM2, Coilin) do not adopt a fixed, folded three-dimensional structure; instead, they possess long disordered, flexible, and highly charged polypeptide chains. These IDRs enable weak and transient multivalent interactions that promote the condensation of macromolecules into liquid “droplets”, separated from the surrounding nucleoplasm without the need for a lipid membrane.
The correlation is direct and fascinating: The same chemical characteristics that enable LLPS (high charge density, repetitions of acidic/basic amino acids, massive phosphorylation) are those that confer intense argyrophilia.
-
Acidic IDRs massively attract silver.
-
The high local concentration of proteins within the condensate (versus the dilute nucleoplasm) amplifies the staining signal.
Therefore, we can state with scientific rigour that Santiago Ramón y Cajal was, inadvertently, the first scientist to systematically map the biomolecular condensates of the nucleus, using silver as a sensor of defined liquid phases characterised by a specific protein composition, where certain zones concentrate proteins with different electronegativity capable of “trapping” silver with different affinity. This perspective revalues his work not merely as descriptive but as a physicochemical detection of the state of nuclear matter.
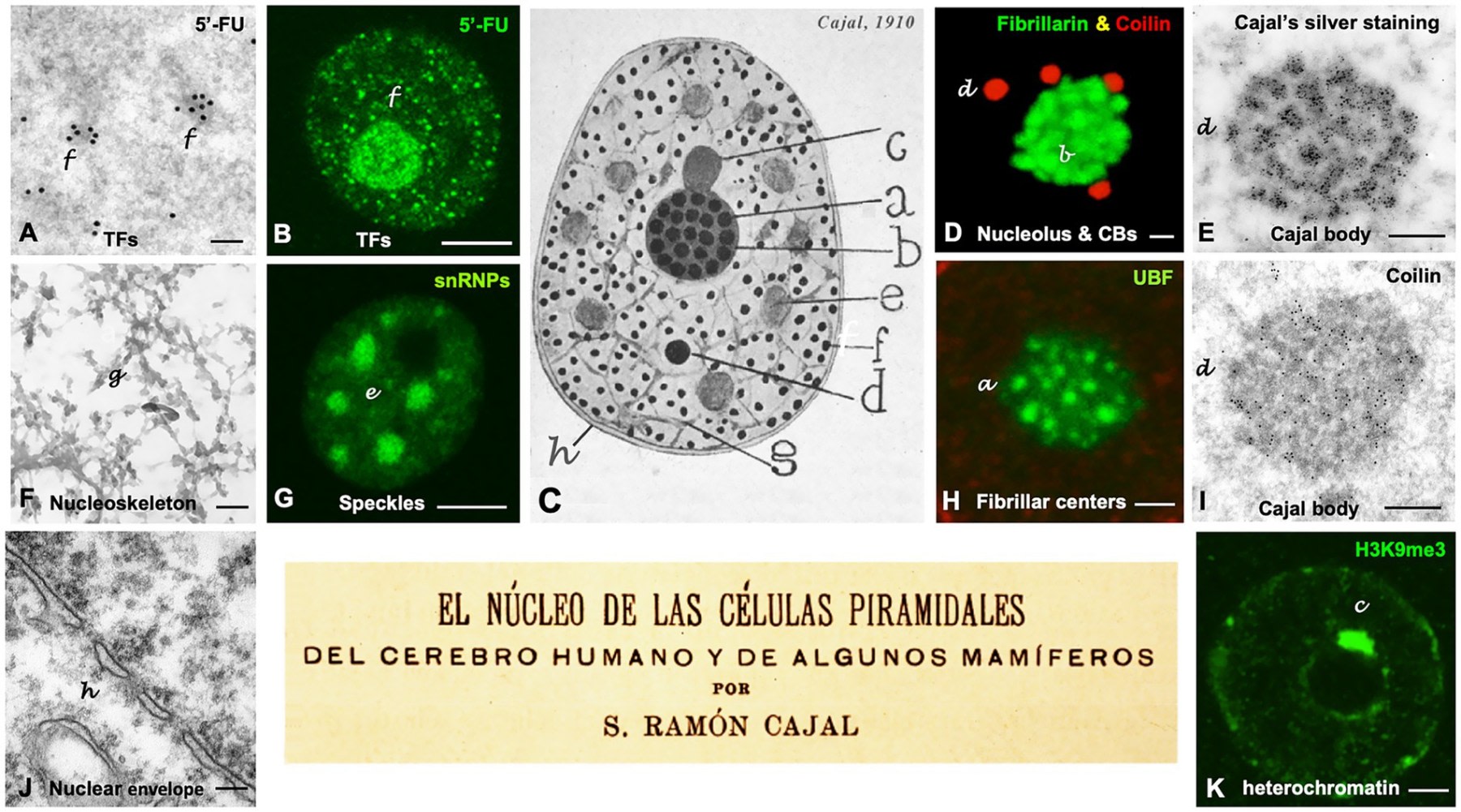
3. The Neuronal Nucleolus: The Ribosome Factory and Its Microphases
The nucleolus is the most prominent structure of the nucleus and was the object of detailed study by Cajal. Far from considering it a homogeneous mass, Cajal described a complex internal architecture composed of argyrophilic spherules densely packed and embedded in a fundamental substance.
3.1 Argyrophilic Spherules: The Equivalence with Fibrillar Centres (FC)
The review by Lafarga et al. establishes an unequivocal correspondence based on correlative microscopy and molecular labelling:
- Cajal’s Argyrophilic Spherules is equivalent to Fibrillar Centre (FC) and Dense Fibrillar Component (DFC) Units.
In modern ultrastructure, the mammalian nucleolus is organised into three concentric functional compartments, representing sequential stages of ribosomal biogenesis:
-
Fibrillar Centre (FC): Contains the rDNA genes (ribosomal DNA) and transcription factors such as UBF and RNA Polymerase I. It is the site of storage and transcriptional pre-initiation.
-
Dense Fibrillar Component (DFC): Surrounds the FCs and is where the active transcription of the 47S pre-rRNA and its early processing (methylation, pseudouridylation) occur. It contains the protein fibrillarin and snoRNPs (small nucleolar RNPs). These processes give rise in eukaryotes to the mature rRNAs: 18S (as part of the small ribosomal subunit), 5.8S, and 28S (as part of the large subunit).
-
Granular Component (GC): The peripheral zone where pre-ribosomal subunits are assembled with ribosomal proteins imported from the cytoplasm. It contains nucleophosmin (B23).
The “argyrophilic spherules” that Cajal drew as black dots correspond to the FC/DFC complexes. Their intense silver staining is due to the very high concentration of UBF and phosphorylated RNA Polymerase I in these regions. Conversely, the silver-refractory “fundamental substance” (stainable with basic dyes or anilines) corresponds to the Granular Component (GC), rich in RNA but with a less argyrophilic protein composition under the specific conditions of Cajal’s technique.
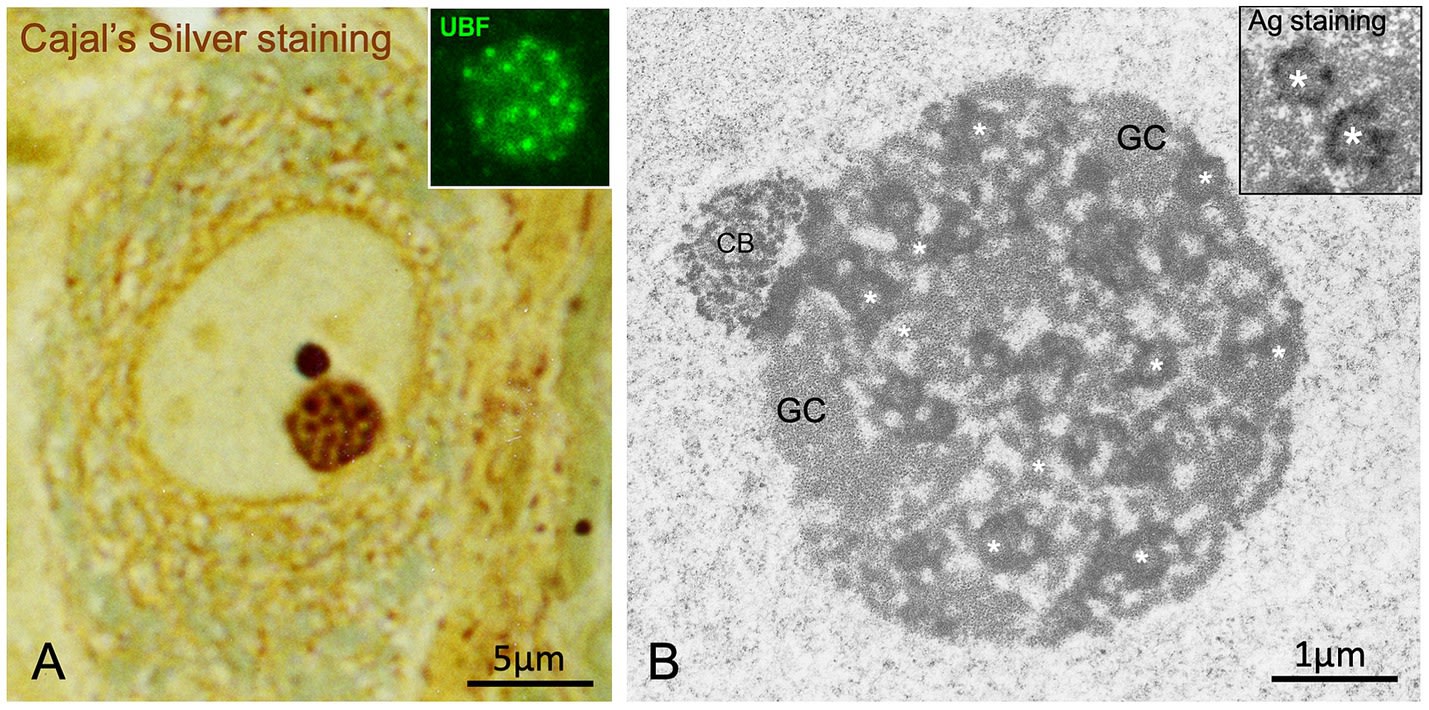
3.2 Nucleolar Dynamics and the Law of Neuronal Size
Cajal not only described the structure but made quantitative observations linking nuclear morphology to cell physiology. He formulated an almost mathematical positive correlation between the size of the neuronal soma and the number of nucleolar argyrophilic spherules:
-
Small Neurons: Presented 4 to 8 spherules.
-
Giant Neurons (e.g. Giant Pyramidal, Motor): Exhibited between 24 and 36 spherules.
This observation constitutes one of the first morphofunctional laws of neuronal cell biology. Lafarga et al. fully validate this “law” in the light of modern molecular biology. The size of a neuron (and the extent of its dendritic and axonal tree) determines its biosynthetic demand. A spinal motor neuron, whose axon can extend up to one metre, requires a massive quantity of proteins to maintain its cytoskeleton, axonal transport, and synaptic function. To satisfy this demand for protein translation, the cell must manufacture ribosomes at a staggering rate.
The increase in the number of argyrophilic spherules (FC/DFC units) reflects the recruitment of more rRNA gene copies (ribosomal genes) for active transcription. The nucleolus, as a liquid condensate, has the capacity to fuse or fission its internal components as needed. In neurons of high metabolic activity, the dispersion of the transcriptional machinery across multiple active foci (many spherules) maximises the contact surface between the polymerase and the genes, optimising rRNA production. Thus, Cajal was visualising, unknowingly, the rate of rDNA transcription and the metabolic state of the neuron.
Furthermore, the article highlights that the nucleolus is not merely a ribosome factory but a stress sensor. Alterations in nucleolar morphology (such as the segregation of its components or the loss of spherules) are early markers of toxicity and neurodegeneration, a concept known today as “nucleolar stress”, implicated in the pathogenesis of diseases such as Parkinson’s and ALS.
4. Hyaline Clumps and Neutrophil Granules: Processing and Transcription
Beyond the nucleolus and the CB, Cajal described more diffuse and numerous nucleoplasmic components, whose identity has been the subject of debate until recent times.
4.1 Nuclear Speckles: Cajal’s “Hyaline Clumps”
Cajal identified irregular structures of translucent (“hyaline”) appearance and moderate silver affinity (staining yellow or reddish), which he termed “hyaline clumps” (“grumos hialinos”). These structures, present in numbers of 6 to 11 per nucleus, were clearly distinguishable from chromatin.
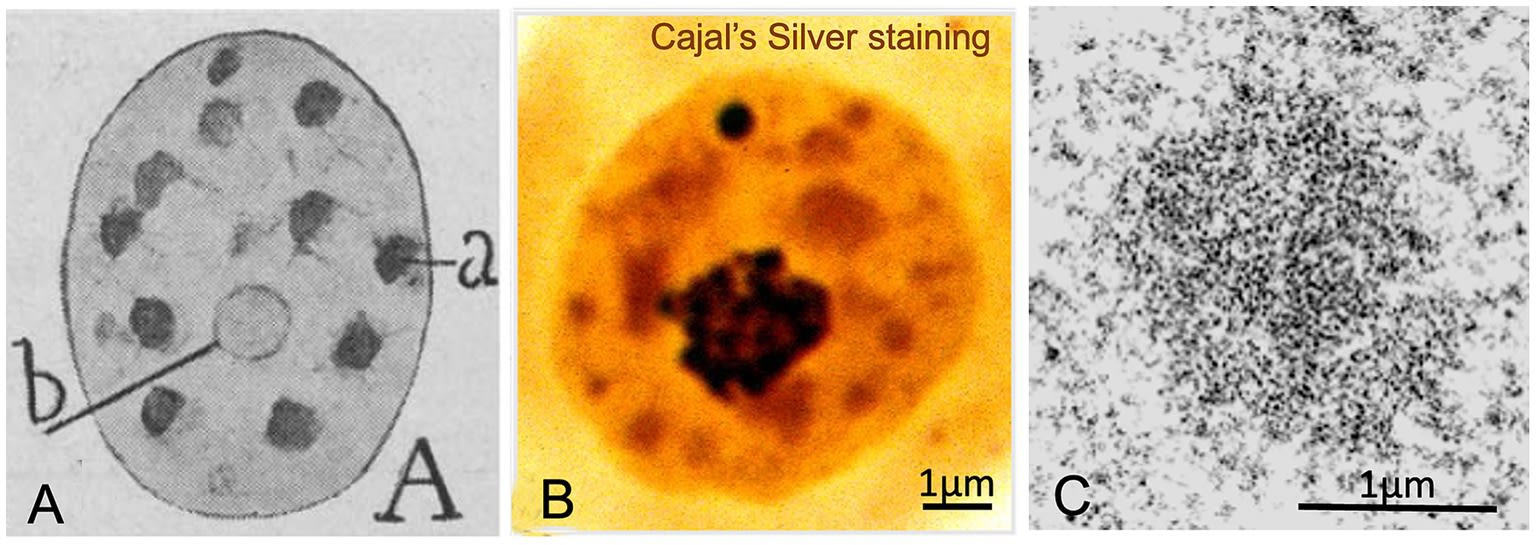
The current review confirms that the hyaline clumps are Nuclear Speckles, also known ultrastructurally as Interchromatin Granule Clusters (IGCs).
-
Molecular Identity: They are domains rich in splicing factors of the SR family (such as SC35/SRSF2) and snRNPs. The protein SRRM2 acts as a fundamental scaffold, containing enormous disordered regions that promote speckle formation via LLPS.
-
Function as Hubs: Speckles are not passive depots. They act as logistics centres. Genes with high transcriptional activity are physically positioned at the periphery of the speckles (zones known as SPADs: Speckle-Associated Domains). This proximity facilitates the immediate transfer of splicing factors to the nascent RNA, spatially coupling transcription and processing.
The alteration of speckles is central to the pathology of Amyotrophic Lateral Sclerosis (ALS) and Frontotemporal Dementia (FTD). In these diseases, the protein TDP-43 (which normally transits through the speckles) and the scaffold protein SRRM2 aberrantly aggregate in the cytoplasm or form insoluble nuclear inclusions. This leads to the “evaporation” or dispersal of functional speckles, provoking a systemic failure in RNA splicing that contributes to neuronal death.
4.2 Transcription Factories (Neutrophil Granules) and the Condensate Controversy
The smallest structures described by Cajal were the “neutrophil granules”: minute dots (0.15 – 0.18 μm) dispersed throughout the nucleoplasm, with a mixed staining affinity and intense punctate argyrophilia.
Lafarga et al. propose that these granules correspond to Transcription Factories (TFs).
-
Evidence: Immunocytochemistry for the active form of RNA Polymerase II (phosphorylated at Serine 2) and RNA precursor incorporation experiments (5-FU) reveal a pattern of active transcription “dots” that coincides in size, number, and distribution with Cajal’s granules.
-
Spatial Organisation: Cajal noted that these granules often surrounded the hyaline clumps (speckles), which fits perfectly with the current model of perispeckle transcription.
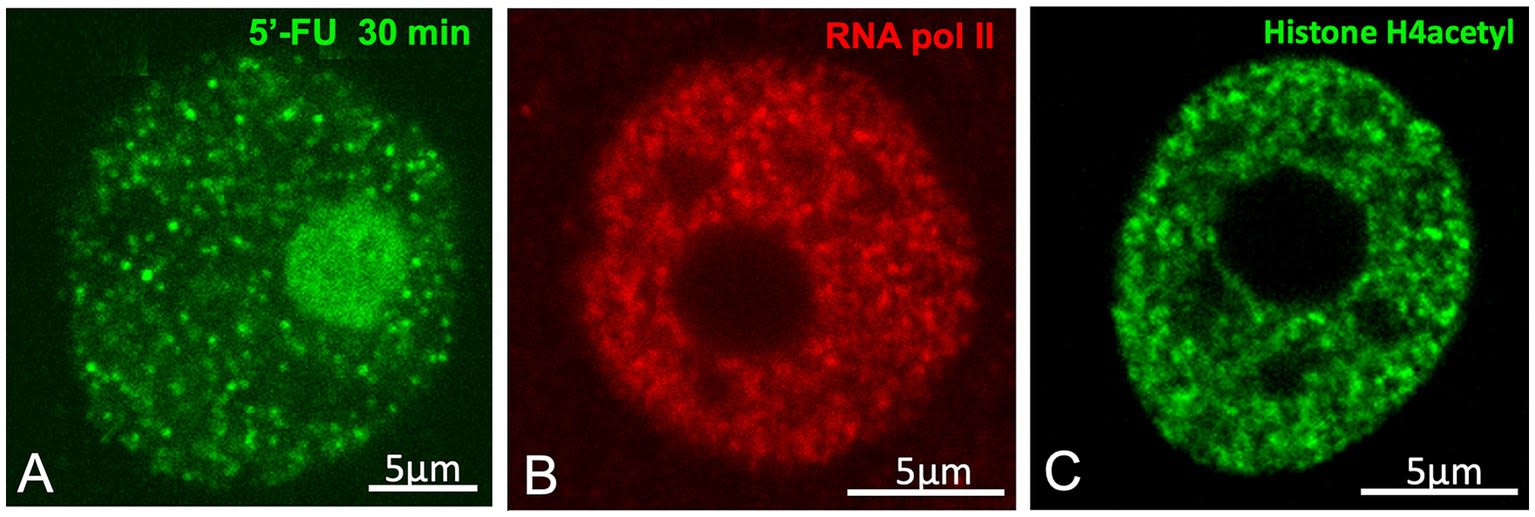
The Stortz et al. (2024) Controversy:
The article does not shy away from the current scientific debate on the physical nature of these factories. Recent works, including those of Stortz et al. (2024), are cited, questioning whether transcription factories are true liquid condensates (formed by LLPS) or simply transient molecular clusters on the DNA (clustering).
-
The Condensate Argument: Holds that TFs are liquid droplets (“Super-enhancers”) that concentrate transcription factors through IDR interactions to cooperatively enhance gene expression.
-
Stortz’s Critique: Argues that evidence for LLPS in vivo for transcription is weak. Suggests that the formation of large condensates could be detrimental (“a curse”) by sequestering factors away from other genes and slowing diffusion, rather than beneficial. Stortz proposes that what we see as “factories” could be a dynamic accumulation without the properties of a phase-separated liquid.
Lafarga et al. integrate this discussion by acknowledging that, while the morphology described by Cajal is indisputable (the “granules” exist as visible entities), the underlying physical mechanism (liquid phase vs. chromatin aggregation) remains an intellectual battleground in 2025.
5. The Cajal Body: From “Accessory Body” to Nuclear Logistics Centre
Among all of Cajal’s nuclear discoveries, perhaps none bears his personal imprint as directly as the “Accessory Body”. Discovered in 1903, this small spherical structure (0.5 – 1.0 μm), frequently attached to the nucleolus, puzzled histologists for decades.
5.1 Identification and Nomenclature: A Hundred-Year Journey
Cajal described the accessory body as a structure with a silver affinity superior even to that of the nucleolus, staining a deep black (“sepia” or charcoal) and displaying great sharpness of contours. Despite his precise description, the structure fell into oblivion or was confused with artefacts until the advent of electron microscopy.
In 1969, Monneron and Bernhard described a nuclear structure composed of coiled threads of high electron density, which they called the “Coiled Body”. It was not until the 1980s and 1990s that the research group of Miguel Lafarga at the University of Cantabria, using ultrastructural silver cytochemistry techniques (a modern adaptation of Cajal’s method), demonstrated that Cajal’s “Accessory Body” and the “Coiled Body” were the same biological entity. Silver precipitates specifically decorated the coiled threads of the body, confirming the structural basis of the argyrophilia observed by Cajal. In recognition of this identity, Joseph Gall proposed in 1999 to rename the structure as the Cajal Body (CB), a name universally accepted today.
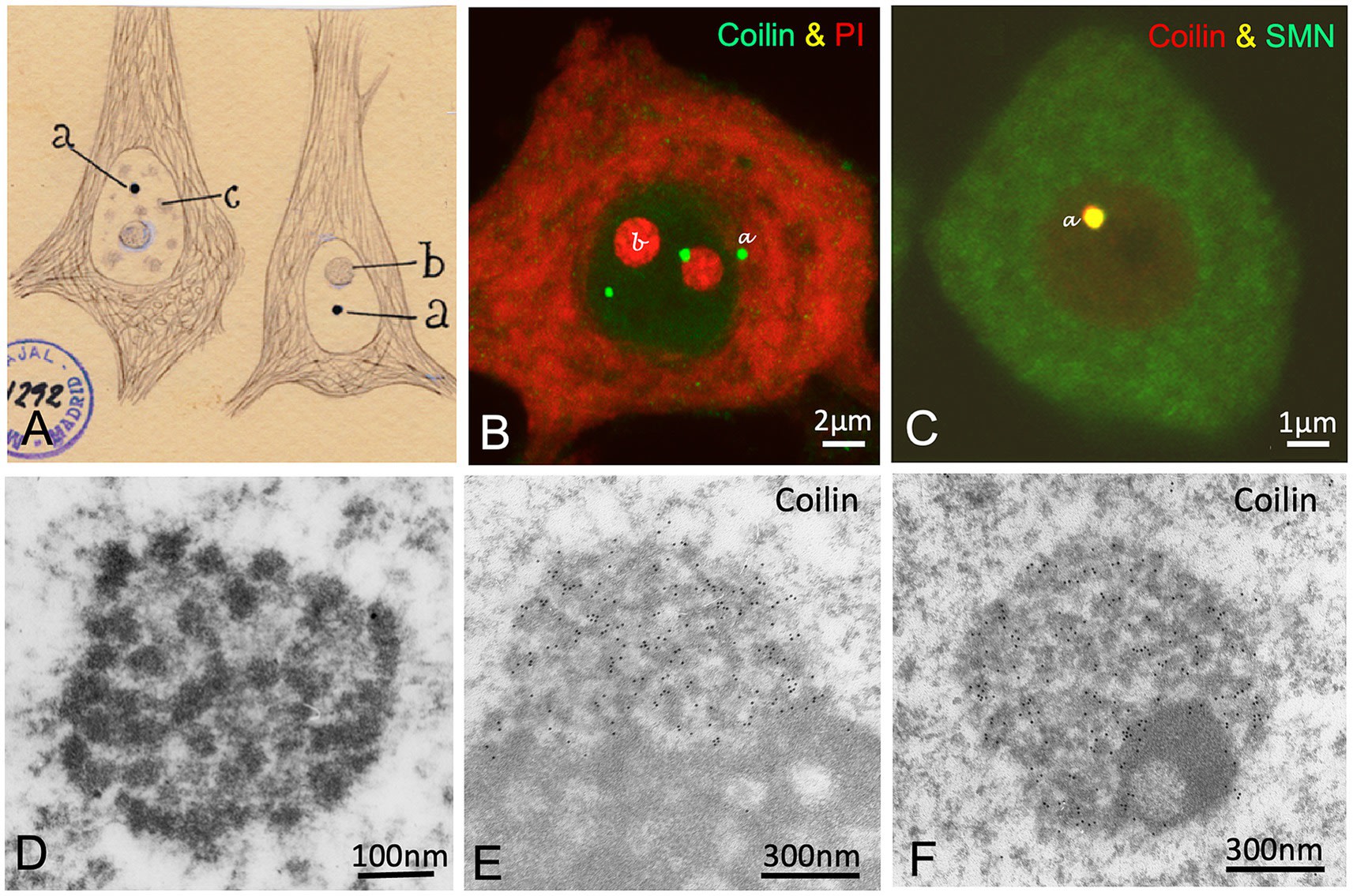
5.2 Molecular Biology of the Cajal Body: Assembly and Maturation
The molecular identity of the CB is defined by the presence of the protein Coilin (p80-coilin), which acts as the molecular scaffold for the body’s assembly. The report by Lafarga et al. details the composition and function of this organelle:
-
Composition: In addition to coilin, CBs concentrate spliceosomal snRNPs (small nuclear Ribonucleoproteins) (U1, U2, U4/U6, U5), snoRNPs (small nucleolar RNPs), the protein SMN (Survival Motor Neuron), fibrillarin, and a specific class of guide RNAs called scaRNAs (small Cajal body-specific RNAs).
-
Function: The CB is not a site of primary transcription but a centre for maturation and recycling. It acts as a “workshop” where immature snRNPs, freshly imported from the cytoplasm, undergo critical chemical modifications (cap methylation, pseudouridylation) guided by scaRNAs. These modifications are essential for the stability and fidelity of snRNPs before they are dispatched to the nuclear speckles to participate in pre-mRNA splicing.
The CB is a paradigmatic example of an organelle dependent on transcriptional activity. Cajal observed that its presence and number varied; today we know that neurons with high transcriptional demand upregulate the number of CBs to ensure a constant supply of mature splicing machinery.
5.3 Pathological Implications: Spinal Muscular Atrophy (SMA)
The clinical relevance of the Cajal Body is dramatic in the context of Spinal Muscular Atrophy (SMA), the most common genetic cause of infant mortality. The article establishes a direct mechanistic connection:
-
SMN Deficiency: SMA is caused by mutations in the SMN1 gene, resulting in low levels of SMN protein.
-
Assembly Failure: SMN is essential for recruiting snRNPs and coilin to the Cajal Body. In the absence of adequate SMN levels, Cajal Bodies do not form or disintegrate in motor neurons.
-
Splicing Collapse: The loss of CBs prevents the efficient maturation of snRNPs. This causes a “bottleneck” in the RNA processing machinery, resulting in massive splicing errors (intron retention, exon skipping) in multiple genes necessary for motor function.
-
Therapeutic Validation: Treatment with Nusinersen (an antisense oligonucleotide that increases the production of functional SMN) has been shown in murine models not only to improve survival but to restore the presence of Cajal Bodies in the nuclei of motor neurons.
This finding is an impressive validation of the functional importance of the structures described by Cajal: their physical presence is an indicator of neuronal health, and their disappearance is a mechanism of disease.
Recommended reading: To delve into a surprising and lesser-known facet of this structure — its role in nuclear immunity and virology — and to discover the connection with Cajal’s facet as a microbiologist, we recommend the article: The Cajal Body.
6. Other Nuclear Structures: The Linin Framework and Levi’s Clump
The review of Cajal’s work reveals that his visual acuity was not limited to the most dynamic condensates; he also described structures that today we know constitute the physical and epigenetic scaffolding of the nucleus. Lafarga et al. (2025) rescue two key components frequently forgotten but which complete the integral vision of the neuronal nucleus.
6.1 The “Linin Framework”: The Nucleoskeleton
Cajal described a “linin framework” (armazón de linina), a trabecular network of filaments connecting the nucleolus to the nuclear membrane. Although historically considered a fixation artefact, modern techniques of high-voltage electron microscopy and resin-free sections have validated this observation. This network corresponds to the nucleoskeleton, and more specifically to the nuclear lamina and its internal projections.
Today we understand that this structure is not merely a passive support. The nuclear lamina organises chromatin into Lamina-Associated Domains (LADs), which generally contain silenced genes. Furthermore, this “framework” is posited to be crucial for nuclear mechanotransduction, allowing the neuron to detect and respond to external mechanical forces by altering its gene expression, a concept that Cajal intuited by drawing the physical continuity between the nuclear interior and the cellular envelope.
6.2 “Levi’s Clump”: Perinucleolar Heterochromatin
In 1910, Cajal also confirmed the presence of a basophilic mass adhered to the nucleolus, originally described by Giuseppe Levi in 1896. Unlike the other argyrophilic structures, “Levi’s Clump” stained intensely with basic dyes (such as toluidine blue) but was refractory to silver.
Modern molecular biology unequivocally identifies Levi’s Clump as condensed perinucleolar heterochromatin, a specific type of Nucleolus-Associated Domains (NADs). This region is enriched in repressive epigenetic marks such as histone H3K9me3. Its function is to maintain the silencing of repetitive genomic regions (such as pericentromeric satellite DNA) and to sequester genes that must not be expressed at a given moment. Cajal’s observation about the variability of this clump between large and small neurons reflects differences in epigenetic architecture and cellular maturation state.
6.3 The Nuclear Envelope: A Double Membrane Visible?
Finally, it is intriguing that both Cajal (1910) and Marinesco (1909) represented the nuclear envelope as composed of a double membrane (Figure 1C), a structural feature that today we clearly identify through electron microscopy (Figure 1J), despite the fact that such a level of detail was theoretically beyond the resolution limit of the optical microscopes of the era.
Lafarga et al. (2025) propose a technical explanation for this “vision”: the apparent double membrane observed by Cajal could have been the result of an excessive dilation of the perinuclear cisterna, probably an artefact caused by the fixation agents used in his reduced silver nitrate method. It is worth noting that Cajal had already drawn the nucleus surrounded by two parallel lines in an earlier study on the large non-neuronal cells of invertebrates (Cajal, 1898), suggesting a consistency in this morphological observation, even if its physical basis was an alteration of the perinuclear space.
7. Comparative Synthesis and Nuclear Pathology
To visualise the magnitude of the convergence between Cajal’s vision and modern science, the following integrative table summarising the article’s findings is presented:
| Structure (Cajal 1910 Nomenclature) | Appearance (Reduced Silver) / Modern Equivalent (2025) / Key Molecular Markers / Formation Mechanism (Proposed) / Associated Pathology (Mechanism) / Argyrophilic Spherules |
|---|---|
| Fibrillar Centres (FC) | and DFC. UBF, Fibrillarin, Pol I. LLPS driven by IDRs of Fibrillarin/Nucleolin |
| Parkinson’s/ALS: | Nucleolar stress, inhibition of protein synthesis. |
| Accessory Body | Very Intense Black (“Sepia”) |
| Cajal Body (CB) | Coilin, SMN, scaRNAs. LLPS mediated by Coilin oligomerisation |
| SMA: | SMN deficit prevents CB formation -> splicing failure. |
| Hyaline Clumps | Yellow/Red (Irregular) |
| Nuclear Speckles | (IGCs). SC35, SRRM2, snRNPs. LLPS mediated by SRRM2 and SON |
| ALS/FTD: | TDP-43/SRRM2 aggregation dissolves speckles. |
| Neutrophil Granules | Black Dots (Dispersed) |
| Transcription Factories | RNA Pol II (Ser2P), Transcription Factors. LLPS or Clustering? (Stortz et al. Controversy) |
| Cancer/Neurodegeneration: | Global transcriptional deregulation. |
| Levi’s Clump | Negative (Basophilic) |
| Perinucleolar Heterochromatin | Histone H3K9me3, HP1. Chromatin condensation (Polymeric phase separation) |
| Ageing: | Epigenetic changes and loss of silencing. |
This table does not merely translate the terminology; it evidences a fundamental principle: Cajal’s argyrophilia acted as a selective filter for biomolecular condensates rich in IDRs and nucleic acids, allowing him to “see” the functional organisation of the genome long before DNA was known.
8. Conclusions and Future Perspectives
The review by Lafarga et al. (2025) is a document that transcends the history of science to become a roadmap for current cellular neurobiology. The conclusions derived from this analysis are profound:
-
Absolute Validity of Cajal’s Model: Cajal’s descriptions from 1910 were not artefacts or erroneous interpretations. They were empirical observations of such precision that, 115 years later, they require only a change of nomenclature to be fully current. Cajal’s “nucleus” is the “modern nucleus”.
-
The Nucleus as a Condensate Ecosystem: The reduced silver technique teaches us that the nucleus is not a dilute solution but an environment densely populated by membraneless organelles. The coincidence between argyrophilia and proteins with IDRs suggests that “nuclear matter” is fundamentally organised through phase separation, a concept that unifies classical histology with cutting-edge biophysics.
-
Nuclear Architecture Is Clinical: The disruption of the structures Cajal drew — the loss of Cajal Bodies in SMA, the dispersal of speckles in ALS — are not mere epiphenomena but direct causes of disease. Restoring the “nuclear geography” (as the drug Nusinersen does with CBs) is a validated therapeutic strategy.
In conclusion, Santiago Ramón y Cajal gave us not only the map of the brain (the connectome), but also the map of the cell’s control centre (the nucleome). In 2025, when we revisit his silver-stained plates, we are not looking at the past; we are finding the keys to curing the diseases of the future. His work remains, in the most literal sense, silvered and brilliant.

Cajal’s organization of neuronal nucleus revisited — Docs.Santiagoramonycajal
El núcleo de las células piramidales del cerebro humano y de algunos mamíferos por S. Ramón Cajal. 990001153000204201_V08 — Docs.Santiagoramonycajal
Manual de Histología normal y Técnica Micrográfica. b21779995 — Docs.Santiagoramonycajal



